The cell motility regulator NAA80 is assisted by Profilin 2
Actin is modified by N-terminal acetylation which regulates its role in steering cellular architecture and cell motility. Now the machinery performing this acetylation is uncovered.

Main content
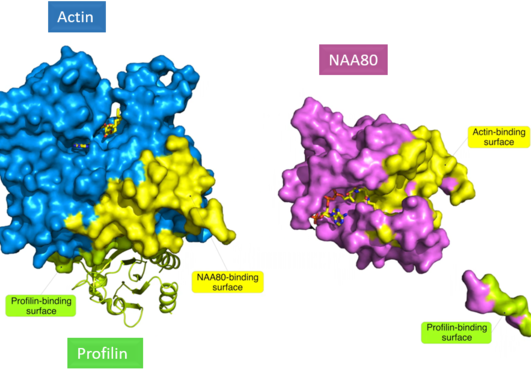
In human cells, actin is a major component of the cytoskeleton. Only recently, the actin N-terminal acetyltransferase NAA80 was identified and NAA80 mediated N-terminal acetylation of actin was revealed to have a strong impact on cytoskeleton dynamics and cell motility.
There is a machinery of seven N-terminal acetyltransferases (NATs) catalyzing N-terminal acetylation of totally 80% of the human proteome. Most NATs therefore have a large number of substrates and many operate on the ribosome to carry out co-translational acetylation. In contrast, actin has a dedicated NAT, NAA80, which acts post-translationally. Important insights on how actin is targeted by NAA80 and profilin was recently revealed by a trimeric structure of actin-NAA80-profilin. Now, a minor isoform of profilin, profilin 2 (PFN2), emerges as the specific cellular partner for NAA80. Binding of PFN2 to NAA80 increases its intrinsic capacity to perform actin N-terminal acetylation and stabilizes the contact between NAA80 and its substrate actin. Thus, the key components defining how actin becomes N-terminally acetylated in human cells are now determined.
Biochemical and molecular biology investigations in the Arnesen lab at UiB were carried out by current lab members Rasmus Ree, Laura Kind and Syvia Varland and alumni Minglu Dai and Adrian Drazic. Analytical ultracentrifugation experiments were performed by Anna Kaziales and Klaus Richter at the Technical University in Munich.
The Arnesen lab acknowledges support from Helse Vest, the Norwegian Cancer Society, the Research Council of Norway and the European Research Council (ERC). Rasmus Ree and Laura Kind are Postdoctoral and doctoral candidates, respectively, supported by the Medical Faculty at UiB.