From ERASMUS internship to published paper
During his Erasmus internship in the Arnesen lab, Tobias B. Beigl took great interest in his project on the recently identified NAA80 enzyme. Beigl was on an interesting and important research track and stayed on for a Master’s project trying to figure out why cells lacking NAA80 typically had a more fragmented Golgi apparatus. He recently published his findings in the scientific journal Experimental Cell Research together with supervisor Henriette Aksnes and colleagues in the research group.

Main content
Tobias joined the Erasmus education exchange programme from the University of Stuttgart and applied for an internship at UiB. He joined the NAT group at a time when they needed to get more knowledge on a recently identified enzyme, NAA80. The NAT group had uncovered NAA80 to be responsible for post-translational modification of actin, a key protein that amongst other things forms the cytoskeleton, a dynamic skeleton in the cells. Actin performs this function through its ability to polymerize into thin threads or filaments. The dynamicity of this cytoskeleton enables cells to migrate. Henriette had previously showed that cells lacking NAA80 migrated faster. Tobias looked at the organelles in these cells and described an increased presence of fragmented Golgi structures in these cultures.
Next, they wondered how fragmentation of the Golgi could be connected to altered actin function. The NAT group researchers joined forces with Prof. Jaakko Saraste who specializes in the functional organization of the secretory pathway, including the Golgi apparatus. During the initial phase of cell migration, the Golgi apparatus actually fragments somewhat in the process of positioning the Golgi favorably for directed migration. Given the hypermotility of NAA80-lacking cells, Tobias then wanted to study the Golgi apparatus of actively migrating cells. He did indeed observe an increased tendency for Golgi fragmentation when the cells were on the move. However, he could still detect a significantly higher number of cells with a fragmented Golgi among the cells lacking NAA80 compared to wildtype cells. This told these researchers that there was something more to this mystery, possibly having to do with an altered actin function since actin’s N-terminus is not normally matured in the absence of NAA80.
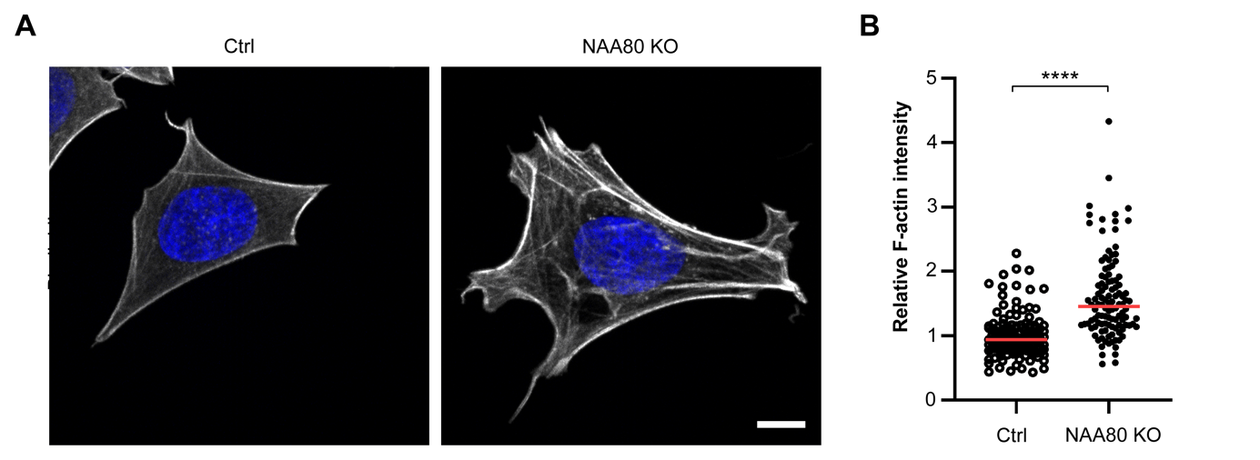
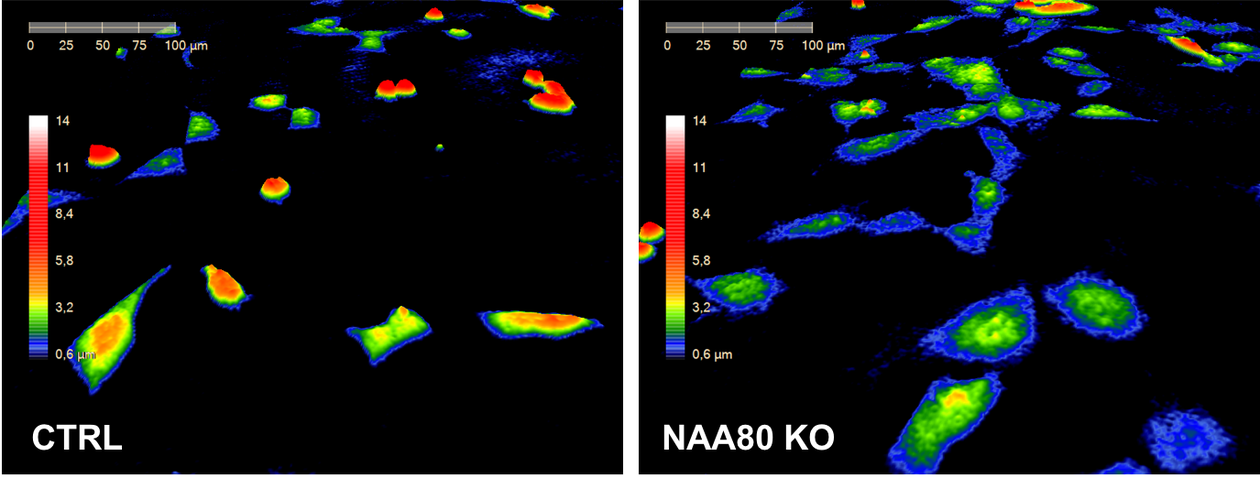
Tobias paired up with NAA80 partner in crime, Monica Hellesvik, also a Master student at the time. Monica had recently optimized an assay for quantification of the amount of filamentous actin by using MIC’s Dragonfly microscope and Imaris analysis software. Thanks to this assay, Tobias and Monica were able to find a possible explanation as to why the Golgi apparatus could take on a less compact form in the cells lacking NAA80. In fact, these cells had much more F-actin compared to normal cells (Figure 1). Increased levels of F-actin was something that had previously been linked to Golgi fragmentation. Monica and Tobi also used the new tool in the NAT group, the HoloMonitor M4, a microscope for live-cell imaging and quantification, to show morphological features of the NAA80-lacking cells that could reflect their increased F-actin content. The HoloMonitor captures images by digital phase holographic microscopy. This technique has greatly facilitated the NAT group’s research and is currently expanding its applications in the hands of Monica Hellesvik who is excited to try the recent software upgrade for the quantitative analysis that is performed on these holographic images (Figure 2).
After graduating with his Master’s thesis June 2019, Tobias started a PhD at the Institute of clinical pharmacology in Stuttgart. Monica and Henriette continue to build on this work and find it fascinating that Golgi fragmentation might be a trait of cancer cells. This is particularly interesting since NAA80 could function as a brake to keep cell migration in check, meaning that lack of NAA80 could possibly facilitate metastasis. Increased fragmentation of the Golgi apparatus in NAA80-lacking cells therefore fits to this hypothesis.
The Arnesen lab acknowledges support from Helse Vest, the Norwegian Cancer Society, the Research Council of Norway and the European Research Council (ERC).
Link to the original article: https://doi.org/10.1016/j.yexcr.2020.111961