Application for animal studies
All animal research in Norway requires approval from the Norwegian Food Safety Authority (Mattilsynet), and applications must be submitted electronically through FOTS – the application and oversight system of the Norwegian Animal Research Authority. On this page, you will find an overview of the requirements, procedures, exemptions, training, and practical information you need to submit a complete and compliant FOTS application.

Main content
Main rule
All persons who plan to conduct experiments involving animals must, according to Norwegian legislation, obtain approval from the Norwegian Food Safety Authority (Mattilsynet). Applications must be submitted electronically through FOTS – the application and oversight system of the Norwegian Animal Research Authority (Forsøksdyrforvaltningen).
We recommend that you read the information below before submitting your application.
Links to more information can be found on the bottom of this page
Application form in FOTS
Applications must be completed and submitted electronically in FOTS.
To obtain access to FOTS, you must contact your local PMSK (Person with Special Responsibility for Control).
What we need when you apply for access to FOTS
- Name
- Work and mobile phone numbers
- Email address
- Position
- Role in projects (project leader/project member)
- Highest level of education
- Completed laboratory animal science course + course certificate
- Previous experience with animal experiments
- Animal species and procedures you have practical experience with
- Documentation of continuous professional development
- Have you had FOTS access before? (Yes/No)
General requirements for a FOTS application
The application must include:
- Documentation of competence for the applicant and everyone involved
- Project summary (NTS)
- Severity classification
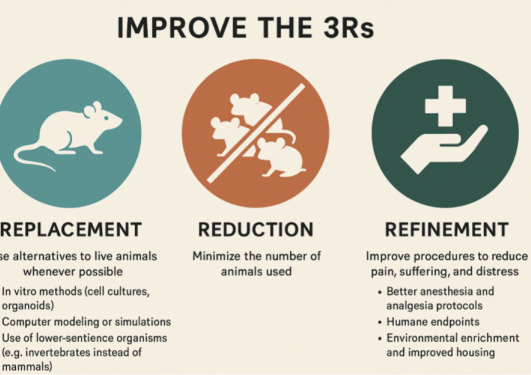
- Explanation of the 3Rs (Replacement, Reduction, Refinement)
- Harm–Benefit assessment
- Number and type of animals
- Funding source
- Planned start and end dates
- Assessment regarding public accessibility
- Background and purpose
- Justification for choice of animal model
- Assessment of phenotype and animal welfare (if relevant)
Description of procedures, including:
- anaesthesia, analgesia, and euthanasia
- preparation of animals
- interventions and handling
- registrations, sampling, monitoring
- humane endpoints
- calculation of animal numbers and group sizes
Nontechnical Project Summary (NTS)
The NTS must be anonymous, easy to understand, and must not contain any confidential information.
It must describe:
- Purpose of the project
- Expected adverse effects on the animals
- Scientific or societal benefits
- Number and type of animals
- How the 3R principles are addressed
All fields in FOTS marked with a globe will be included in the NTS
The summary is published by Mattilsynet and in the EU database ALURES.
Processing time
Applications to the Norwegian Animal Research Authority (FDF) in Mattilsynet are processed as quickly as possible, and no later than 40 or 55 working days, depending on the complexity of the project.
Training requirements
Everyone performing animal experiments must complete mandatory training in laboratory animal science.
Amendments to approved projects
Amendments must be submitted in FOTS:
- Changes that do not negatively affect animal welfare: submit as a notification
- All other changes: submit an application for changes
The project summary must always be updated.
Guidance from MAttilsynet
Mattilsynet’s official guidance for using FOTS
Workflow
When an application is submitted, it is first reviewed by:
- The local Person with Special Responsibility for Control (PMSK)
- The Animal Welfare Unit (DVE)
For the application to be processed, the invoice address must be complete and include a PM number (or Merida number for the hospital).
Fees
Mattilsynet has established fixed fees for processing FOTS applications.
Exemptions from application requirement
§ 6 – Euthanasia before use
No application is required when animals are only euthanized using approved methods (as listed in Annex C of the Regulation on the Use of Animals in Experiments) and organs/tissues are used for research.
This exemption applies only to the application requirement.All other regulatory requirements—competence, housing, routines—still apply.
This applies to all animals bred for scientific purposes, regardless of origin.
Breeding exempt from application
Breeding is exempt from application if the animals:
- do not have a harmful phenotype*, and
- identification/tissue sampling is performed using one ear biopsy.
To determine whether a phenotype is harmful, a systematic assessment must be carried out:
- 10 animals of each sex
- from at least 2 different litters
- observed until 40 weeks of age
The animal facility has a dedicated protocol, which is assessed by a veterinarian.
* Immunodeficient phenotypes are considered harmful, and breeding/keeping such animals requires a FOTS application.
If the veterinarian concludes that the phenotype is not harmful, a simplified notification may be submitted instead of a full application.
Forms:
- Phenotype assessment protocol – send to vetpost@viv.uib.no
- Simplified notification – send to your local PMSK
Other questions
Other questions about FOTS can be submitted to Aurora Brønstad