News archive for Structural neurobiology
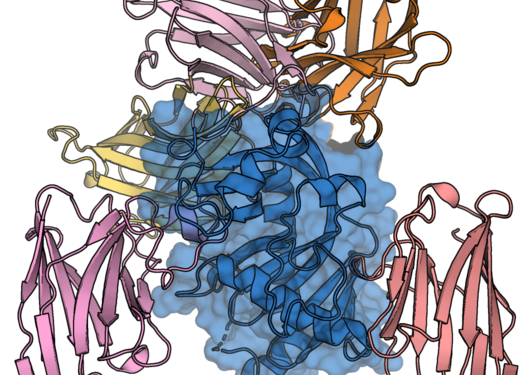
CNPase is an enzyme present at high levels in myelinating cells. Novel nanobody tools developed in an international collaboration were used to identify binding epitopes at atomic resolution and visualise myelin. Additionally, the nanobodies were used as intrabodies to bind to CNPase in living cells. These nanotools will be valuable for future research on myelin and its molecules.
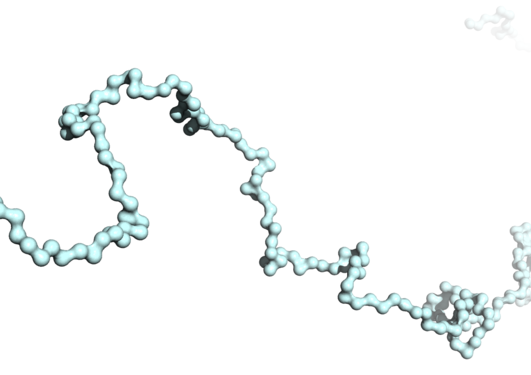
Myelin is a crucial structure for the normal functioning of the vertebrate nervous system. Here, we have used multiple state-of-the-art technologies at large international research infrastructures, enabling methods not available in Norway, to obtain 3D structural information on some of the most hydrophobic protein molecules in the human body, which are responsible for the correct structure and... Read more
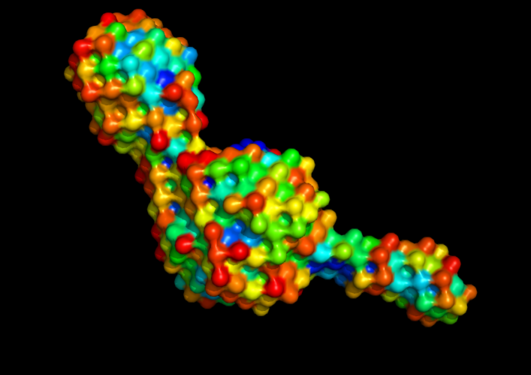
Nanobodies are single-domain fragments obtained from camelid antibodies, which have high affinity towards their target protein. Two recent publications describe the development and use of nanobodies directed against the neuronal Arc protein to promote both structure determination, imaging and detection, and functional studies.
Malaria is one of the most devastating infectious diseases in the world. The parasites causing malaria move by gliding, for which force is generated by an unusual actomyosin motor. We have determined a high-resolution crystal structure of one of the key regulators of parasite actin dynamics, the homodimeric actin capping protein alpha.
Our research project BIOPROM (Biomimetic Proteolipid Multilayers - Structure and Properties) was awarded a research grant of 12 MNOK by the Research Council of Norway. The BIOPROM project will reveal unprecedented details of the molecular structure and properties of myelin, from the nano to the meso scale.
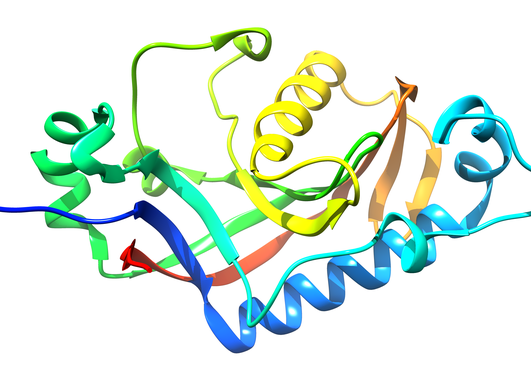
Charcot-Marie-Tooth disease (CMT) is the most common inherited neuropathy in humans, linked to mutations in dozens of different genes. Two of these genes encode the proteins GDAP1 and NDRG1. We have solved the crystal structures of these two proteins, in order to better understand their structure, function, and involvement in disease.
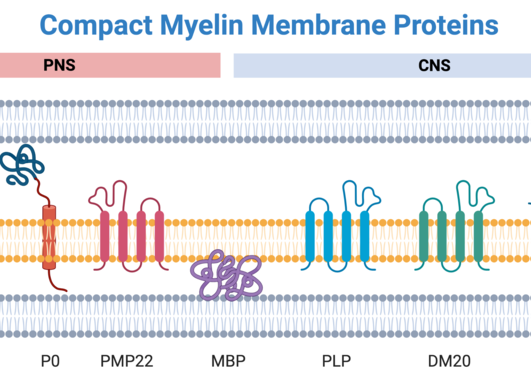
Myelin, the insulative multilamellar sheath that enwraps axons, speeds up our nerve impulses by two orders of magnitude – a prerequisite for an efficient nervous system that we humans and other vertebrates enjoy. Myelin gains its structure and function from a high abundance of lipids and proteins, many of which are specific to this enigmatic biological system.
The ILL brochures are meant for the general public with a scientific interest, and they cover the application of neutron techniques in different areas of research. The "Neutrons in Health" brochure is published every 5 years, and 20 topics are selected globally from top research performed at the ILL (Grenoble, France). Two of these 20 highlights in the latest issue are from our work on the... Read more
One long-standing question in myelin biochemistry was solved, as a new binding partner specific for the large isoform of MAG was identified.
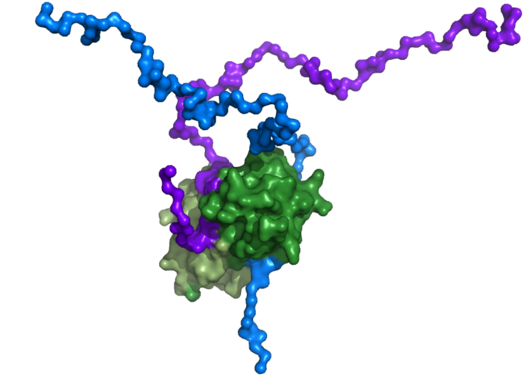
The post-synaptic protein Arc has been termed a master regulator of memory. In line with its homology to certain viral proteins, it has also been shown recently to form virus-like capsids. We have elucidated for the first time the 3D structure of full-length monomeric Arc.
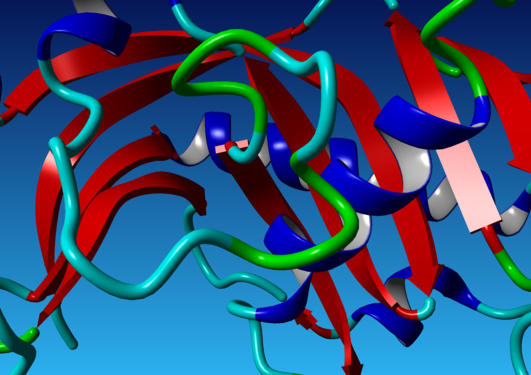
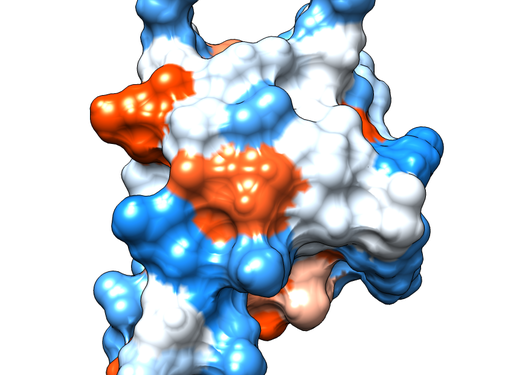
The Shank family of proteins functions as a molecular scaffold in the neuronal post-synaptic density, enabling numerous protein-protein interactions. Shanks are large multi-domain molecules, and one of the conserved domains is an SH3 domain. Using high-resolution X-ray crystallography, we show that the SH3 domain in the Shank family has lost its canonical ligand-binding site.
The molecular processes of myelin maturation are to a large extent still unknown. A coordinated interplay between regulatory and functional molecules must take place for correct timing of myelin membrane apposition and compaction, such that cytoplasmic channels are also formed in a correct way.
- December 2024 (1)
- September 2022 (1)
- June 2022 (1)
- November 2021 (1)
- September 2021 (1)
- February 2021 (1)
- February 2020 (1)
- November 2019 (1)
- December 2018 (1)
- August 2018 (1)
- June 2017 (1)
- January 2017 (1)